Number of Atoms in CaSO4 (Calcium sulfate) |

|
|
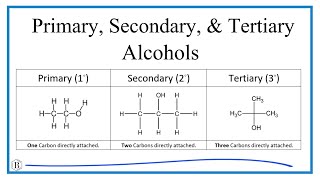
To find the number of atoms in CaSO4, Calcium sulfate, we’ll add up the number of atoms for Ca, S, and O. The small number after the element symbol is called the subscript and this tells us how many of that atom are in the compound.
When there is no number after an element, as is the case with Calcium (Ca), we assume there to be one atom of that element. For example, in CaSO4 we have only one Ca atom. For CaSO4 there are a total of 6 atoms in the compound. To find the number of atoms in one mole of CaSO4 we multiply the number of each atom (for example there are four Oxygen atoms) by Avogadro's number, 6.02 x 10^23. |