How to Convert Moles of MgO to Grams |

|
|
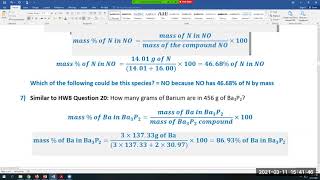
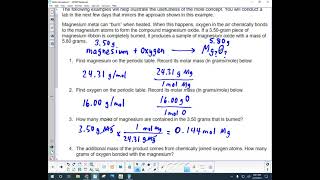
In this video well learn to convert moles of MgO to grams. The technique used can be applied to any mole to gram conversion. For our practice problem we’ll convert 1.39 mole of MgO (Magnesium oxide) to grams. Using the same process in reverse we could also convert grams to moles.
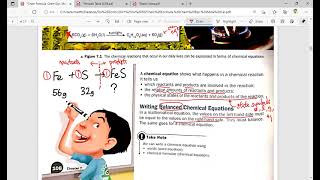
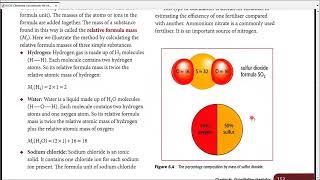
First, we'll find the molar mass of MgO. After that we'll use the mole map (https://www.breslyn.org/chemistry/stoich/Printable-Mole-Map.php) to find the grams of MgO in 1.39 moles of Magnesium oxide (MgO). For more help with moles to grams conversions and more: • More Moles to Grams Practice: https://youtu.be/aIv5nr8ZNyw • Molar Mass in Three Easy Steps: https://youtu.be/o3MMBO8WxjY • Understanding the Mole: https://youtu.be/DyLktMPTuHY • Moles - Gram Conversions: https://youtu.be/aIv5nr8ZNyw • How to Balance Chemical Equations: https://youtu.be/zmdxMlb88Fs • Mole Ratio: https://youtu.be/i71BMVlrMiw • Reaction Stoichiometry: https://youtu.be/rrTqOsZPpaU My chemistry website: http://www.Breslyn.org Converting between moles and grams is the cornerstone of being successful in stoichiometry, the study of chemical quantities. Take the time to learn mole conversions and you will find chemistry is much easier. The use of conversion factors (also called factor-label method or dimensional analysis) is a more general technique for converting quantities. Once you understand how it works it can be applied to many different conversion (as long as you know the conversion factor). |












![[Chemistry] Problem Solving Session #2](https://ytimg.googleusercontent.com/vi/D2jSvJyklK0/mqdefault.jpg)