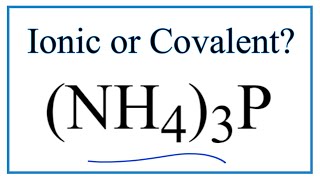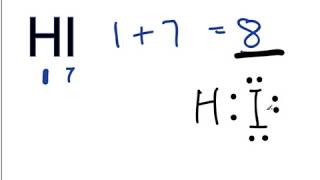Is HI Ionic or Covalent/Molecular? |

|
|
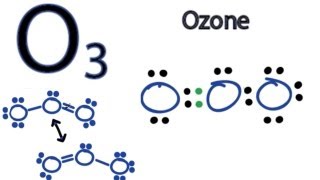
To tell if HI (Hydrogen iodide or Hydroiodic acid) is ionic or covalent (also called molecular) we look at the Periodic Table that and see that H is non-metal and Br is a non-metal. When we have a non-metal and a non-metal the compound is usually considered covalent.
For HI there will is a larger difference in electronegativity between the non-metal and non-metal. This difference results in an electron(s) being shared unequally with the non-metal having a higher electronegativity having the electron more of the time.. --- Helpful Resources Metals, Non-Metals on the P- Table: https://youtu.be/OoooStZQHdA Ionic, Covalent, & Polar Covalent: https://youtu.be/OHFGXfWB_r4 Electronegativity for each element: https://en.wikipedia.org/wiki/Electronegativity --- Because we have a combination of a non-metal and non-metal HI (Hydrogen iodide or Hydroiodic acid) is considered an covalent compound. For more chemistry help, see http://www.Breslyn.org. |