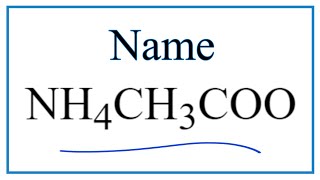
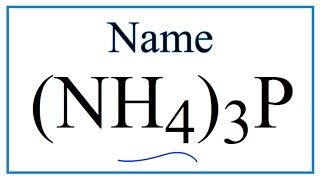
How to Write the Name for Al2(SO4)3 |

|
|
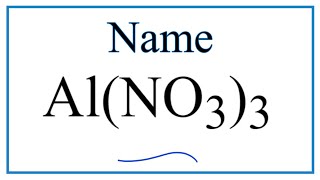
In this video we'll write the correct name for Al2(SO4)3. To write the name for Al2(SO4)3 we’ll use the Periodic Table and follow some simple rules.
Because Al2(SO4)3 has a polyatomic ion we’ll also need to use a table of names for common polyatomic ions, in addition to the Periodic Table. Common Ion Table: https://breslyn.org/chemistry/naming/resources/common_ion_table.php ---Keys for Naming Ternary Ionic Compounds--- 1) Name the metal (the cation) as it appears on the Periodic Table. Na+ = Sodium Mg2+ = Magnesium Al3+ = Aluminum 2) Find the polyatomic ion on the Common Ion Table and write the name. Note: It is possible to have two polyatomic ions such as NH4NO3. In this case find and write both names as found on the Common Ion Table. ----------------------------- For a complete tutorial on naming and formula writing for compounds, like Aluminum sulfate and more, visit: http://www.breslyn.org/chemistry/naming Drawing/writing done in InkScape. Screen capture done with Camtasia Studio 4.0. Done on a Dell Dimension laptop computer with a Wacom digital tablet (Bamboo). |