🧪How Do Bases React With Metals?🧪 [H2O + NaOH + Al] | Class 10 Science Chapter 2 | Learn Practically |

|
|
In this video, I have explained what happens when bases react with metals; the chemical reaction of H2O + NaOH + Al (Water + Sodium Hydroxide + Aluminium) will take place to produce Sodium Aluminate with the evolution of Hydrogen Gas and Heat Energy
The video concludes that Metal reacts with bases to produce Salt and Hydrogen Gas with a rise in temperature (evolution of heat energy). BUT, unlike acids, not every base reacts with metals in a chemical reaction. Complete video link - https://youtu.be/CzsD6xGQP7Q Metal + Base → Salt + Hydrogen Gas . . . #Class10 #Science #Chemicalreaction |
![🧪How Do Bases React With Metals?🧪 [H2O + NaOH + Al] | Class 10 Science Chapter 2 | Learn Practically](https://ytimg.googleusercontent.com/vi/dOLhU_x7AkU/mqdefault.jpg)
![🧪How Do Acids React With Metals?🧪 [HCl + Mg] | Class 10 Science Chapter 2 | Learn Practically](https://ytimg.googleusercontent.com/vi/UTTSie9ujis/mqdefault.jpg)

![Extracting Salt [MgCl2] 🧪How Do Acids React With Metals?🧪 Class 10 Science Chapter 2](https://ytimg.googleusercontent.com/vi/ZlhkmKGKM4Y/mqdefault.jpg)

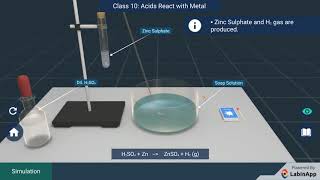










![🧪How Do Acids React With Metals?🧪 [HCl + Fe] | Class 10 Science Chapter 2 | Learn Practically](https://ytimg.googleusercontent.com/vi/m4_OUnznDdM/mqdefault.jpg)




![🧪How Do Acids React With Metals?🧪 [HCl + Al] | Class 10 Science Chapter 2 | Learn Practically](https://ytimg.googleusercontent.com/vi/xY6_uL4AFm4/mqdefault.jpg)






